Take Home Messages
Barley Grain Production
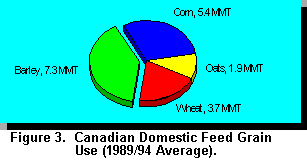 Barley makes up 40% of feed grain usage in
Canada which is equivalent to 7.3 MMT compared to 5.4 MMT for corn (Figure 3). Barley is the
primary feed grain used in beef and dairy cattle diets in western Canada. Historically, the price
of
barley has tracked the price of corn quite closely, although the corn price was significantly higher
for much of 1993 and 1994.
Barley makes up 40% of feed grain usage in
Canada which is equivalent to 7.3 MMT compared to 5.4 MMT for corn (Figure 3). Barley is the
primary feed grain used in beef and dairy cattle diets in western Canada. Historically, the price
of
barley has tracked the price of corn quite closely, although the corn price was significantly higher
for much of 1993 and 1994.Nutrient Composition of Barley and Other Feed Grains
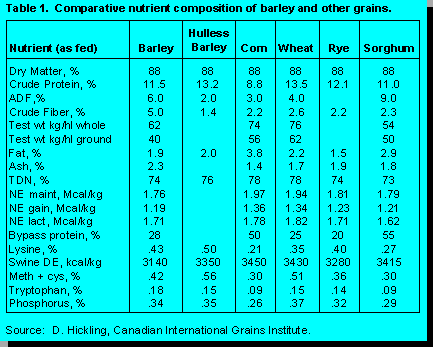
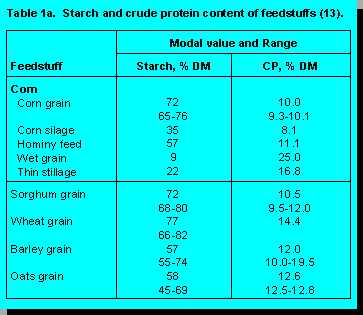 Hulless barley has received considerable
attention in recent years, primarily as a feedstuff for non-ruminants. However, interest in the use
of
hulless barley in dairy cattle diets is also increasing as it becomes more widely available.
Although acreage sown to hulless barley is quite modest, it is increasing rapidly from 88
thousand
acres in 1993/94 to an estimated 110 thousand acres in 1994/95. Because the proportion of hull
is
much lower, hulless barley has a higher concentration of protein and a lower fiber concentration
than hulled varieties. Its energy value for swine approaches that of barley plus it has the added
advantage of being a superior source of amino acids. Information on the feeding of hulless
barley
to dairy cattle is currently not available. One of the concerns with respect to feeding hulless
barley
to ruminants is that its lower fiber level may be associated with more rapid digestion in the rumen
and thus result in increased incidence of digestive disorders such as acidosis. A key factor here
is
whether selection of hulless varieties has resulted in any changes in the rate and extent of starch
digestion in the rumen. This raises several questions. What should the selection criteria be for
barley? Is it logical to have the same selection criteria for ruminants as non-ruminants? Do we
need specialized barley varieties for forage production? Are the selection criteria being applied
to
malting varieties compatible with high nutritional quality for ruminants? The market for barley as
a grain source for ruminants is sufficiently large to justify a selection program targeted
specifically
to the needs of ruminants. The task is not an easy one as it will require agreement among
ruminant nutritionists on the most appropriate selection criteria.
Hulless barley has received considerable
attention in recent years, primarily as a feedstuff for non-ruminants. However, interest in the use
of
hulless barley in dairy cattle diets is also increasing as it becomes more widely available.
Although acreage sown to hulless barley is quite modest, it is increasing rapidly from 88
thousand
acres in 1993/94 to an estimated 110 thousand acres in 1994/95. Because the proportion of hull
is
much lower, hulless barley has a higher concentration of protein and a lower fiber concentration
than hulled varieties. Its energy value for swine approaches that of barley plus it has the added
advantage of being a superior source of amino acids. Information on the feeding of hulless
barley
to dairy cattle is currently not available. One of the concerns with respect to feeding hulless
barley
to ruminants is that its lower fiber level may be associated with more rapid digestion in the rumen
and thus result in increased incidence of digestive disorders such as acidosis. A key factor here
is
whether selection of hulless varieties has resulted in any changes in the rate and extent of starch
digestion in the rumen. This raises several questions. What should the selection criteria be for
barley? Is it logical to have the same selection criteria for ruminants as non-ruminants? Do we
need specialized barley varieties for forage production? Are the selection criteria being applied
to
malting varieties compatible with high nutritional quality for ruminants? The market for barley as
a grain source for ruminants is sufficiently large to justify a selection program targeted
specifically
to the needs of ruminants. The task is not an easy one as it will require agreement among
ruminant nutritionists on the most appropriate selection criteria.
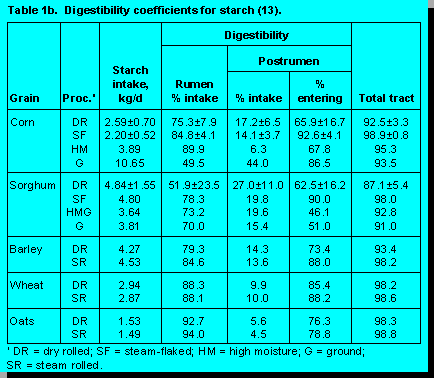
The rumen is the first site of starch digestion by its varied population of resident microorganisms. The extent and amount of starch digested in the rumen, the types and proportion of end products formed and fermentation rates vary and are influenced by 1) grain type, 2) method of cereal processing, 3) other components of the diet, and 4) passage rate (Table 1b, 17, 27). Total tract digestibility of starch across grain sources is usually in the high nineties (91 to 99%) indicating some compensatory effect of postruminal digestion. This compensatory effect is more pronounced for corn and sorghum and less so for barley and wheat (13). For a more comprehensive review of sites of starch digestion the reader is referred to Okine and Kennelly (27).
 Cereal grains are composed primarily of
starch (~60%) and the rate of starch digestion can be a major factor influencing the incidence of
digestive disorders, abscessed livers, and overall productivity of the animal. If digestion of starch
occurs too rapidly, pH drops, microbial digestion is inhibited, feed intake is reduced, and animals
may go off feed. Corn starch is digested relatively slowly in the rumen and thus is known to be
less prone to cause digestive disorders, whereas, barley and wheat starch digestion is quite
rapid.
Cereal grains are composed primarily of
starch (~60%) and the rate of starch digestion can be a major factor influencing the incidence of
digestive disorders, abscessed livers, and overall productivity of the animal. If digestion of starch
occurs too rapidly, pH drops, microbial digestion is inhibited, feed intake is reduced, and animals
may go off feed. Corn starch is digested relatively slowly in the rumen and thus is known to be
less prone to cause digestive disorders, whereas, barley and wheat starch digestion is quite
rapid.
Triticale is a cereal grain developed from wheat and rye. Ruminal digestion of triticale appears to be greater than for barley (12). Ruminal DM disappearance (digestion) for corn, wheat, and three different cultivars of barley (Figure 4) were measured in a recent study in our laboratory. The observed differences in DM disappearance for the various barley cultivars indicates that significant genetic diversity may exist for this characteristic.
Microbial Digestion of Starch. The rumen is inhabited by a dynamic microbial population which number on the average 1010 cells per gram of rumen digesta (17). The composition of the microbial flora changes in response to diet, intake levels, feeding frequency, diurnal variation, and time after feeding. Bacteria adhere to and colonize grain particles in the rumen and hydrolyze the �-1-4 and �-1-6 bonds of the two major molecules of starch, amylose, and amylopectin. It seems that there is a succession of saccharolytic bacteria with streptococcus species increasing two to three times in number within hours of feeding, protozoan species are eliminated and there is a dominance of lactobacilli within 24 hours when high grain diets are fed.
Ruminal protozoa aid in the starch digestion process by ingesting and digesting starch particles. Indeed, the amylolytic protozoan population varies with type of diet, feed intake, and individual animal. The ciliates predominate in grain-fed animals and exert their influence on starch digestion by ingesting bacteria in sufficient quantities to reduce the rate of rumen fermentation and/or by ingesting starch particles and thereby decreasing the accessibility of these substrates to fermentation by bacteria. The rate and extent of ruminal digestion of starch is higher when protozoa are eliminated from the rumen (22).
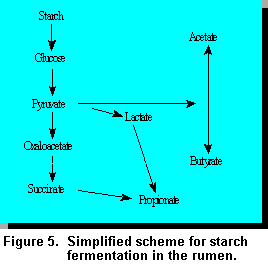 The simple sugars (glucose) released by the
amylolytic bacteria are rapidly fermented to the volatile fatty acids (VFAs), lactate, and carbon
dioxide among other gases (Figure 5). In general, the higher the soluble fraction, or the more
accessible the starch, the higher the rate of starch digestion in the rumen (27).
The simple sugars (glucose) released by the
amylolytic bacteria are rapidly fermented to the volatile fatty acids (VFAs), lactate, and carbon
dioxide among other gases (Figure 5). In general, the higher the soluble fraction, or the more
accessible the starch, the higher the rate of starch digestion in the rumen (27).
Apart from the obvious adverse effects associated with rapid fermentation of high grain diets such as bloat, lactic acidosis, and abscessed livers, there appears to be very little overall advantage to the ruminant associated with the proportion of VFAs produced (27). On the other hand, the readily available energy from starch digestion in the rumen may induce an increase in the amount of microbial protein leaving the rumen (32, 40). Increased microbial protein flow from the rumen to the small intestine reduces the need for ruminal escape protein and it also provides an opportunity for the animal to utilize amino acids as a source of glucose in situations where glucose availability is limiting production. However, any microbial starch reaching the small intestine lowers apparent digestibility of grain starch in the rumen.
Lactic Acidosis. Lactic acidosis is one of the adverse effects associated with rapid fermentation of high grain diets which leads to decreased productivity and occasionally is fatal. The microorganisms involved are principally Streptococcus bovis and lactobaccili. These organisms convert glucose to lactate, but obtain only a relatively small (~2 ATP) amount of energy compared with the 4 or 6 ATP obtained by other microbes which convert glucose to the major VFAs (Figure 5). Under normal circumstances, the lactate producing bacteria exist in very small numbers in the rumen. However, as the amount of starch in the rumen increases, the lactate producing bacteria begin to predominate since they have a greater capacity to absorb and digest the soluble sugars and they also have a lower doubling time. In essence, there is a lowering of rumen pH (increased acidity) which kills the methanogens, cellulolytic, protozoa and lactate utilizers. These processes then cause an increase in osmotic pressure of the rumen leading to entry of water into the rumen and consequently diarrhea, decreased blood levels, hemoconcentration, lameness, bloat, and in some cases, death.
Intestinal Digestion of Starch
Although it is now accepted that dependent on type of grain, up to 50% of dietary starch can escape the rumen, the fate of that starch in the small intestine is still controversial. In this review, special attention is placed on amylase secretion because the efficiency of starch digestion in the small intestine is of great economic concern in ruminants (8). Indeed, the theoretical advantage of absorbing glucose as an end product of starch digestion in the small intestine compared to fermentation products in the rumen (31) created a major research impetus into the capacity of the small intestine to digest, absorb, and transport starch and/or glucose. The question has been whether there is a limitation to starch digestion, absorption, transport, and even metabolism in the small intestine. It has been suggested that the capacity of the small intestine to digest starch could be low due to limited enzymatic hydrolysis of starch. The pancreas produces and releases digestive enzymes in a well defined manner regulated by both neural and hormonal signals (8). However, the role of pancreatic amylase in the utilization of escaped starch by ruminants has been rigorously debated (30, 31). Owens et al. (31) concluded that the form of dietary starch reaching the small intestine was the limiting factor determining starch utilization, and that amylase secretion is adequate. In addition, rskov (30) among others have suggested that pancreatic amylase and/or intestinal maltase may be limiting leading to inadequate digestion of starch to glucose. Although these concepts have not been proven to the satisfaction of everybody, it is becoming entrenched that there is an upper limit of starch digestion in the small intestine of about 1 kg/d for growing cattle and 1.3 kg/d for dairy cattle (13, 18). However, ruminant nutritionists have not adequately examined several issues relating to the role of pancreatic amylase in intestinal starch digestion.
Recent work by Taniguchi et al. (35, 36) showed that there is increased digestibility of starch and increased net appearance of glucose in the portal vein of steers in direct response to increased bypass protein supply to the small intestine. These results are evidence of the notion espoused by Okine and Kennelly (27) that the capacity of the cow to digest starch has been underrated and that the inadequate starch digestion reported by other workers could be explained by lack of adequate protein in the small intestine. As suggested by Fuskiki and Twai (11) and reviewed by Okine and Kennelly (27) an adequate protein level in the small intestine is needed and is essential for the release of starch digesting enzymes from the pancreas. In essence, a high dietary CP level may spare the enzyme releasing hormone (cholecystokinin-releasing peptide) from digestion by trypsin. This allows the releasing peptide to stimulate cholecystokinin release from the intestinal wall which then stimulates amylase release. On the other hand, a low level of protein in the small intestine does not alleviate the inhibition of cholecystokinin-releasing peptide by trypsin and thus inhibits amylase release. Furthermore, an extensive review of the literature by Nocek and Tamminga (26) supports the view that the cow can and does digest substantial amounts of starch in the small intestine.
Production Responses as Influenced by Grain Source
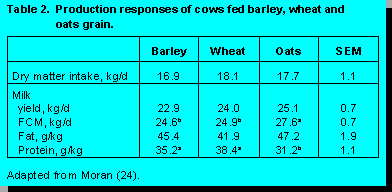 Comparison of cereal grains in terms
of their influence on animal performance have produced mixed results. Tommervik and
Waldern (37) and Jeffery et al. (14) both reported similar yields of FCM with barley,
wheat, and oats whereas Moss and Prier (25) and Moran (24) reported a higher milk yield
of FCM for cows fed oats than cows fed barley and wheat (Table 2).
Comparison of cereal grains in terms
of their influence on animal performance have produced mixed results. Tommervik and
Waldern (37) and Jeffery et al. (14) both reported similar yields of FCM with barley,
wheat, and oats whereas Moss and Prier (25) and Moran (24) reported a higher milk yield
of FCM for cows fed oats than cows fed barley and wheat (Table 2).Grain source may affect rumen fermentation patterns, thus affecting milk composition. Cows fed oats had slightly higher milk fat and lower protein than cows fed wheat or barley. These changes in milk composition were reflections of changes in rumen fermentation. Rumen pH values were 6.98, 6.88, and 6.69 for cows fed oat, barley, and wheat, respectively, while the ratios of acetic + butyric to propionic acid were 4.17, 3.84, and 3.24, respectively (24).
In a study conducted at the University of Alberta barley was evaluated relative to corn in terms of its effect on intake, milk yield, and milk composition. The comparison between barley and corn also included an evaluation of the effect of the dietary source of protein. Thus, barley and corn were fed with both a rapid and a slowly digested protein source (Table 3). Performance of animals were very similar regardless of the source of grain or protein in the diet. Interestingly, source of grain influenced the composition of milk fat; cows fed barley had higher concentrations of C16 and lower concentrations of C18:1.
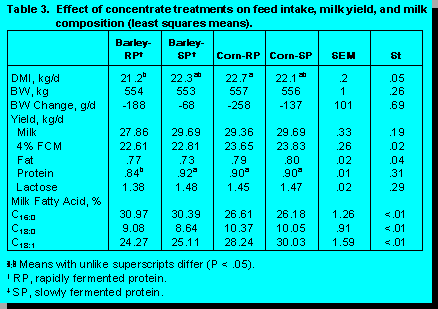
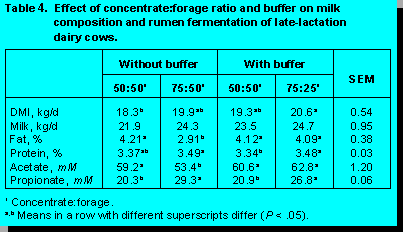 The results indicate the extent to which
milk fat content can be altered by dietary manipulation, range 2.9 to 4.2%. The addition of a
buffer (sodium bicarbonate) completely reversed the effect of the high grain diet on milk fat
percentage.
The results indicate the extent to which
milk fat content can be altered by dietary manipulation, range 2.9 to 4.2%. The addition of a
buffer (sodium bicarbonate) completely reversed the effect of the high grain diet on milk fat
percentage.
With lactating cows the response to cereal grains also depends on the physical form of the grains. Physical processing of cereal grains improves their digestion (19). However, the degree of improvement depends on the grain source. Sorghum and, to a lesser extent, corn benefit more from processing than barley, wheat or oats.
As the rate of ruminal starch fermentation is higher for barley than corn, starch degradation may influence the rate of ammonia utilization by altering the supply of energy for microbial growth (34). However, two studies with lactating cows fed corn or barley as starch sources did not show a clear response to ruminal synchronization of energy and protein (15, 20). It appears that barley may influence milk composition more than milk yield when it is used to replace corn in dairy cattle (Table 3).
Valentine and Wickes (39) fed barley in three forms (whole, rolled, or alkali-treated) and noted the lowest milk yield with whole barley, and higher and similar yields with rolled or alkali-treated barley. rskov et al. (29) noted with oats that alkali-treated barley improved utilization to a greater degree than coarse rolling whereas with barley, utilization was greater with the rolled grain. Clearly, there are differences among grain types in the way that cattle respond to physical or chemical processing.
Selecting Improved Barley Varieties for Ruminants
Dr. Helm and his group at Alberta Agriculture Crop Centre in Lacombe have worked with researchers at the University of Alberta to identify barley cultivars with relatively slow rates of ruminal digestion. From the University of Alberta, Lehman et al. (unpublished data) reported that in situ rumen degradation rate for barley DM digestion of 22 different cultivars ranged from 25 to 35% and these differences could not be related to the location where barley cultivars were grown. Khorasani et al. (unpublished data) evaluated 60 cultivars of barley and observed a range in in situ DM degradation rates of 20 to 62% /h. The observed variability in the ruminal digestion characteristics of barley cultivars indicates that genetic selection holds promise as a means of enhancing the nutritional quality of barley for ruminants.
Who Decides Varietal Selection Criteria for Feed Barley?
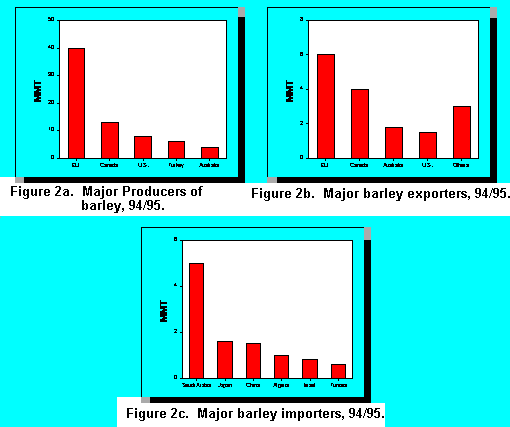
Although the malting trade accounted for only 17% of the barley trade it has a major influence on the barley varieties which are actually used as a feed source for animals. A large percentage of the total barley acreage is planted to malting varieties as farmers play the annual malting lottery. The significant price differential between malting and feed barley encourages grain farmers to plant malting varieties in the hope of meeting the malting specifications. Thus, much of the barley fed to livestock is malting barley which failed to meet the specifications for malting. To the extent that the criteria to be met for malting are not consistent with that needed for animal feed, e.g. relatively lower protein content; this is a major obstacle to the improvement of barley as an animal feedstuff. This has been particularly true in recent years when the price of feed barley has been depressed. As long as a major differential exists between the malting and feed barley price, it is unlikely that this situation will change markedly in the foreseeable future.
Agronomic Factors Drive Varietal Selection
To be successful plant breeders must introduce new varieties which have satisfactory yields and which are relatively resistant to disease. Thus, these and other agronomic characteristics drive varietal selection and there has been relatively little emphasis on the impact of these selection criteria on the nutritional value for the end user, the animal. Animal nutritionists must share the blame for this situation as we have not provided clear direction to plant breeders with respect to the desirable qualities of a feed barley for livestock. It may well be that it is unrealistic to expect nutritionists to speak with a unified voice as the characteristics of the ideal feed barley will differ depending on whether it is fed to swine, poultry, or cattle.
The Cow, the Pig, and the Chicken
A plant breeder interested in improving the nutritive value of barley has to deal with the question: Does selection for traits of interest to swine and poultry nutritionists also enhance the nutritive value of barley for ruminants? Clearly, the fiber content of barley is of much greater concern to non-ruminants than ruminants. In spite of its higher fiber content the energy value of barley for ruminants is relatively close to that for corn and performance may not differ due to source of grain. In contrast, the energy value of barley for swine is significantly less than for corn. However, hulless barley is similar to corn in terms of its digestible energy content. Is hulless barley a suitable feed for ruminants? The answer right now is clearly we do not know. We could speculate that reducing the amount of hull could exacerbate the problems associated with the rapid digestion of barley in the rumen. Unless of course the starch in hulless varieties differs in the rate and extent of ruminal digestion.
Clearly we have a problem if we insist on attempting to satisfy the needs of cattle, swine, and poultry in a single selection program. Is it time to start developing specialized varieties for ruminants which take into account the fermentation characteristics of the varieties being selected?
Whole Plant Barley as a Forage Source
Composition of Cereal Forages
Forage is the key resource for milk production and it accounts for up to 65% of feed for dairy cattle; thus the quality of forage is an important determinant of cattle performance. To determine the optimal quantity and type of forage to include in balanced dairy rations, consideration should be given to nutrient content of forage (e.g. energy and protein), and the digestibility of forage cell walls (NDF). Fiber content is an important measure of forage quality. Lactating dairy cows require a minimum amount of effective fiber in the diet to maintain a healthy rumen. On the other hand, too much fiber in the diet can reduce DMI through increased rumen fill (16, 23, Khorasani et al., unpublished data). The relationship between dietary NDF concentration and characteristics of diets that limit intake, such as bulk density, rumen fill, particle size distribution, digestibility, and rate of digestion and passage from the rumen are important aspects of forage quality.
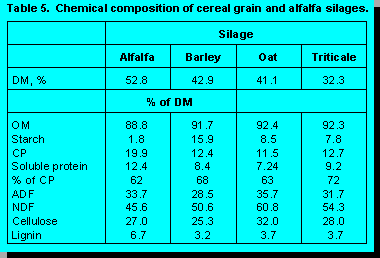 We recently completed an experiment where
we compared the feeding quality and nutrient composition of cereal grain silages with alfalfa
silage. As expected, the protein content of alfalfa silage was higher than that of the cereal grain
silages, which averaged 12% of DM (Table 5); differences among cereal silages were small.
Triticale silage contained the highest soluble protein content, barley silage was intermediate, and
alfalfa and oats silage were lowest in soluble protein content.
We recently completed an experiment where
we compared the feeding quality and nutrient composition of cereal grain silages with alfalfa
silage. As expected, the protein content of alfalfa silage was higher than that of the cereal grain
silages, which averaged 12% of DM (Table 5); differences among cereal silages were small.
Triticale silage contained the highest soluble protein content, barley silage was intermediate, and
alfalfa and oats silage were lowest in soluble protein content.
Alfalfa silage contained higher ADF and lignin, and lower NDF concentration than the cereal grain silages. Within cereal grain silages, oat had the highest ADF and NDF concentration, triticale was intermediate, and barley was lowest (Table 7). Christensen (6) reported that cereal grain silages usually contain 52 to 58% NDF in the DM, but that alfalfa forages seldom contain more than 46% NDF. In our studies (16, Khorasani et al., unpublished data) we reported that cows fed silage with high NDF content (oat and triticale) had lower DMI than cows fed silage with relatively lower NDF (alfalfa and barley). Based on NDF content the quality of the cereal grain silages were ranked in the order of barley, triticale, and oats.
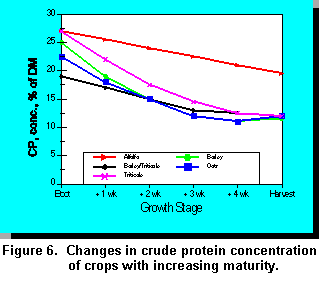
Cereal Forage Maturity and Quality
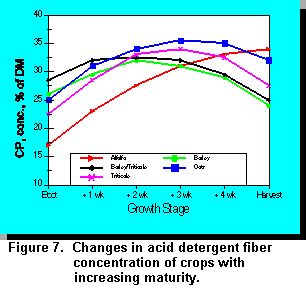
Stage of maturity at harvest significantly affects the yield, protein, and fiber content of cereal forages. Forage fiber content in turn has a significant effect on intake and milk production. Dry matter yields of cereal forages usually increase up to the soft-dough stage of maturity (5, 38) whereas CP concentration decreases with advancing maturity (Figure 6). Acid detergent fiber (Figure 7), cell wall constituents (CWC), and lignin concentrations increase with pre-heading maturation, but remain constant after heading (5). The relationship between stage of maturity and NDF concentration differs among some cereal forages which may influence the optimal time of harvest. Fearon et al. (10) reported that after tillering, the digestibility of triticale decreased with advancing maturity. Baron et al. (1) reported that in vitro digestible organic mater (IVDOM) of barley remained constant over a seven week period after heading, indicating that IVDOM content cannot be used as the sole criterion for determining the optimal harvest date. Baron et al. (1) reported that maximum whole-plant yield is achieved when whole-plant barley is harvested at 42% DM; harvesting at 30% DM reduced yield by about 17%. Cherney and Martin (5) reported that barley had higher in vitro digestibility and lower CWC, ADF, and ADL than oats, wheat, or triticale.
A number of production studies have been conducted at the University of Alberta to evaluate cereal forage quality. The performance data for two studies in early lactation and one study in mid-lactation cows are presented in Tables 6, 7 and 8.
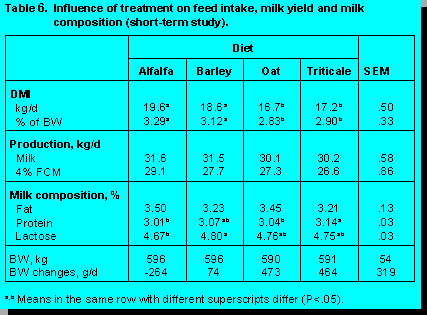
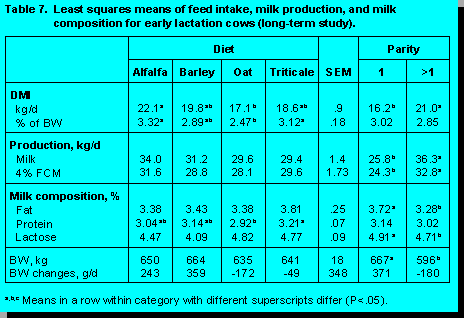
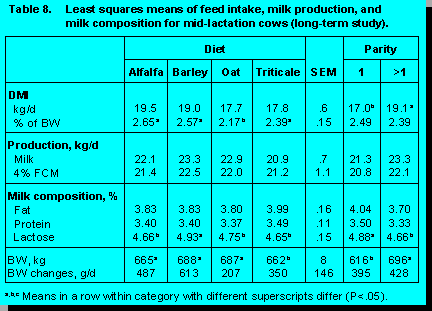
In the University of Alberta studies there were no differences among diets in milk yield, 4% fat corrected milk (FCM), milk energy output, and milk fat percentage. In early lactation, milk from cows fed triticale silage contained a higher milk protein percentage than cows fed the other diets, whereas, silage type had no effect on milk protein concentration in mid-lactation cows. Burgess et al. (2) observed similar milk protein concentrations for cows fed oat, barley, and wheat silage, but they reported a lower milk fat percentage for cows fed wheat silage, with no differences between barley and oat silage. The lower milk fat concentration in cows fed wheat silage may be a reflection of the substantially lower ADF concentrations observed for wheat silage.
Performance of steers fed cereal grain silage have been studied by various investigators. Oltjen and Bolsen (28) found that steers fed oat silage gain at about half the rate as those given barley silage (0.49 versus 1.06 kg/d). These researchers also reported similar performance for barley and corn silage, whereas feeding oat silage resulted in reduced animal performance. McCartney and Vaage (21) reported a higher final body weight and average daily gain for heifers fed barley silage, intermediate for heifers fed oat silage and lowest for heifers fed triticale silage. On the other hand, ZoBell et al. (41) found that barley and triticale silages produced similar body weight, average daily gain, and feed efficiency when fed with barley grain at relatively low levels (26 to 38% of the diet). The reduced animal performance with oat and triticale in steers were related to higher NDF content, lower rate of ruminal digestion, and whole tract digestibility of nutrients.
Exploiting Genetic Diversity to Improve Cereal Forage Quality
 The apparent genetic diversity among barley
cultivars potentially provides an opportunity for selection on the basis of characteristics
considered
to be important for ruminants. Digestible energy content is one of the most important
characteristics of a forage. Forage digestibility influences growth rates of young livestock and
milk production of lactating animals. By affecting passage rate in the digestive tract of animals,
digestibility also influences the amount of forage animals will consume. Genetic diversity seems
to exist in forage species for concentrations of NDF, ADF, lignin, cellulose, and hemicellulose.
Genetic and cultivar variation for cell wall digestibility is often related to cell wall lignin
concentration. Variation in cell wall lignin concentration among cultivars of orchardgrass and
smooth bromegrass was reported by Buxton (3) and Buxton and Russell (4). Little is known
about the genetic variation for specific compounds in cell walls of cereal grain forages. We
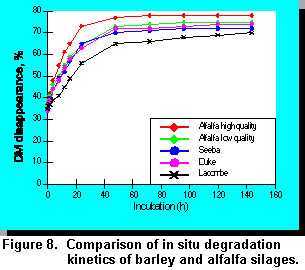
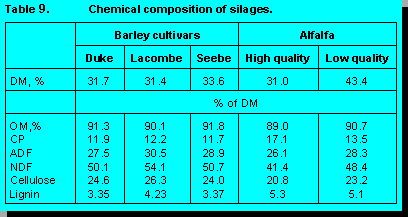
conducted an experiment to compare the ruminal digestibility of three cultivars of barley (Duke,
Lacombe, and Seebe). The chemical composition of these barley silages are presented in Table
9.
Low and high quality alfalfa silage were also included for comparison. Our preliminary results on
ruminal digestion of DM are presented in Figure 8. Data in Table 9 indicate that differences
among barley cultivars in this experiment were relatively minor. Further research is required to
determine the extent of genetic variance among other barley cultivars. However, this task will not
be easy due to the difficulty in controlling factors such as stage of maturity and environmental
effects.
The apparent genetic diversity among barley
cultivars potentially provides an opportunity for selection on the basis of characteristics
considered
to be important for ruminants. Digestible energy content is one of the most important
characteristics of a forage. Forage digestibility influences growth rates of young livestock and
milk production of lactating animals. By affecting passage rate in the digestive tract of animals,
digestibility also influences the amount of forage animals will consume. Genetic diversity seems
to exist in forage species for concentrations of NDF, ADF, lignin, cellulose, and hemicellulose.
Genetic and cultivar variation for cell wall digestibility is often related to cell wall lignin
concentration. Variation in cell wall lignin concentration among cultivars of orchardgrass and
smooth bromegrass was reported by Buxton (3) and Buxton and Russell (4). Little is known
about the genetic variation for specific compounds in cell walls of cereal grain forages. We
conducted an experiment to compare the ruminal digestibility of three cultivars of barley (Duke,
Lacombe, and Seebe). The chemical composition of these barley silages are presented in Table
9.
Low and high quality alfalfa silage were also included for comparison. Our preliminary results on
ruminal digestion of DM are presented in Figure 8. Data in Table 9 indicate that differences
among barley cultivars in this experiment were relatively minor. Further research is required to
determine the extent of genetic variance among other barley cultivars. However, this task will not
be easy due to the difficulty in controlling factors such as stage of maturity and environmental
effects.